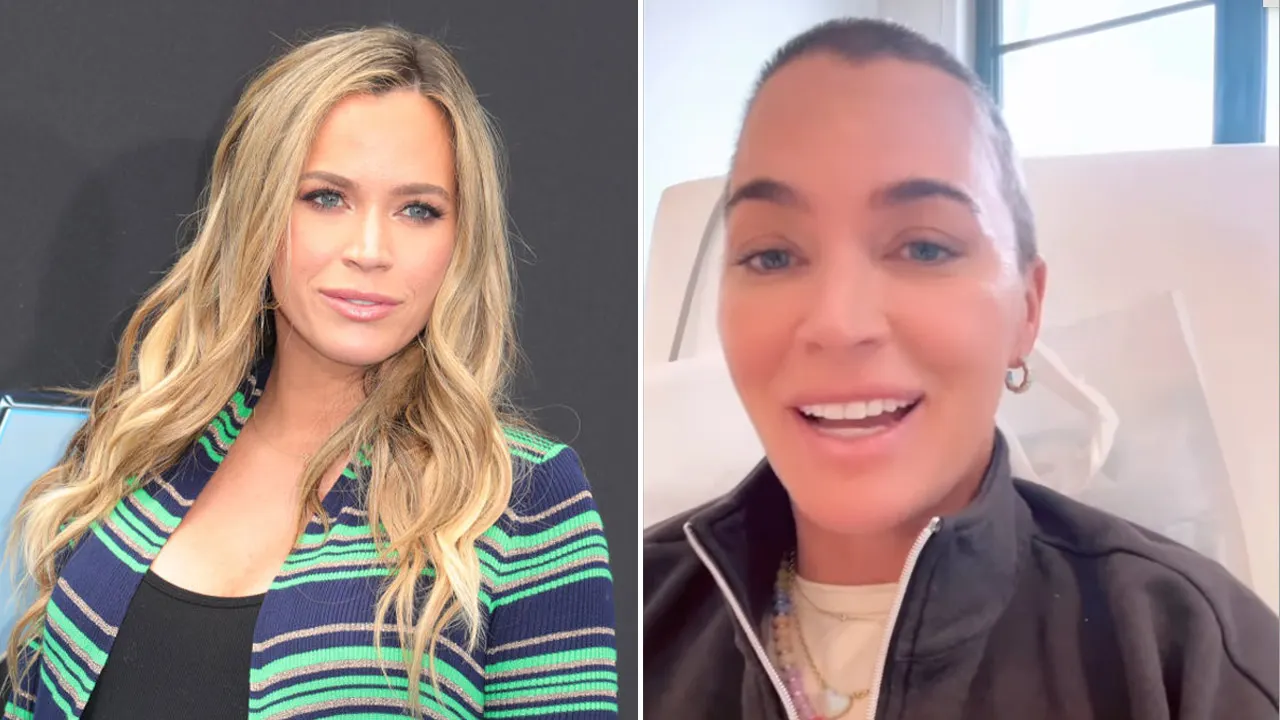
'My daughter has an ultra-rare brain tumour that made her drink 5 litres of water per day'
A teenage girl found herself drinking five litres of water daily due to an "extreme thirst" that couldn't be quenched. Despite drinking more than double the recommended amount, Alissa Bowman, now 17, still felt parched. Initially suspecting …